INTRODUCTION
Incisional hernia is the most common complication of laparotomy. Two million laparotomies are performed annually in the United States, with 200,000 patients requiring incisional hernia repair each year (1); patients underwent hernia repair with suture experienced a zthreefold increase in hernia recurrence rates when compared with patients who had a repair with meshes (2)
The 10-year incisional hernia recurrence rate is reported to be 63% for traditional suture repair without mesh and 32% for repairs using prosthetic mesh. (3) There is currently no ideal mesh, and surgeons must choose the ‘best’ available. There is a rising demand for materials to replace or augment a patient’s native tissue, mesh has become an indispensable tool in hernia repair to improve outcomes and reduce costs. Reducing the complications of mesh implantation is of ultimate importance, most meshes currently used, are nontoxic, stable, chemically and physically inert and non-immunogenic.
Mesh:
There is still controversy about which mesh to use in any specific surgery. Wound healing is influenced by patient’s comorbidities, genetic factors, and dynamic of the abdominal wall, that can contribute to hernia formation. There are more than 70 types of meshes commercially available.
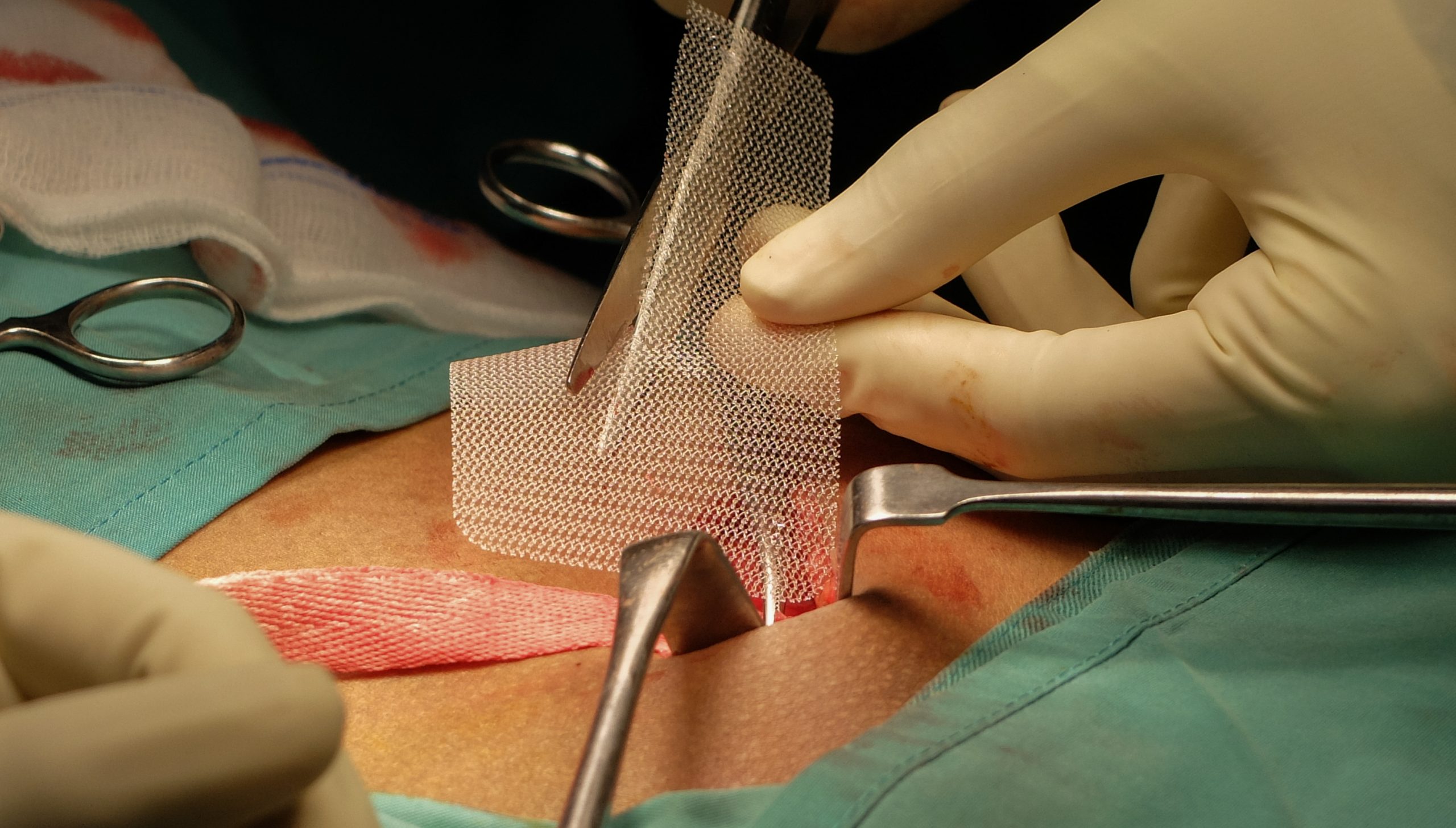
The porosity of mesh plays an important role in outcomes. It has a key role in the reaction of the tissue to the mesh, bacterial growth and cell proliferation are highly dependent upon porosity and pore size, meshes that have large pores have shown to enable entry of macrophages, fibroblasts and collagen that will establish into the new connective tissue, the prosthesis easily integrate to the organism and prevent the biofilm.
Rising demand for materials to replace the native patient tissue became even more complex with the introduction of the biosynthetic meshes in addition to the already existing synthetic and biologic meshes, biosynthetic meshes were developed as a possible cost-effective alternative to the biologic meshes.
Mesh products can be divided in two groups, synthetic and biologic. Synthetic can be further divided into subgroups with and without a barrier, synthetic-partially absorbable, and biological materials.
Synthetic nonabsorbable, are generally the least expensive, made with material as: Polypropylene, Polyethylene polyester and polytetrafluoroethylene. These materials are made in a variety of forms, either with mono or multifilament, They come in a variety of densities and sizes, the optimal density and porosity remain unknown(4). This type of mesh “shrinks” approximately 40% and may vary with the density, filament size and pore size. Pre-placement calculations are needed by the surgeon to achieve a correct fitting and avoid recurrences.
The development of absorbable and biologic meshes were triggered by the complications of using permanent meshes (5). Synthetic degradable mesh, Phasix® and Phasix ST® has been made with ingredients intended to reduce adhesions and provide a safe alternative for use in infected fields. Components of this mesh are Polyglactin, Polyglycolic acid, and trimethylene carbonate, Poly-4-hydroxybutyrate, and Polyglycolide/ trimethylene carbonate /polylactide. Depending on the material it could take from 1 month up to 2 years for mesh to degrade.
Biologic mesh is an Animal-derived tissue, that has been processed to be suitable for use as an implanted device. Derived from biologic decellularized (human, bovine, or porcine) sources, these mesh theoretically incorporate into native tissue and possess the ability to resist infection (6), because of this , they are routinely used in infected fields, their high costs remain a barrier to widespread use.(7) The cumulative data regarding biologic mesh under contaminated conditions do not support claims that is better than synthetic mesh. Biologic mesh use should be avoided when bridging is needed. In inguinal hernia repair biologic and biosynthetic meshes do not have a clear advantage over the synthetic meshes.
New developments in mesh design talk about nanofibers, which show a strong potential to be used in the next generation of surgical mesh, different materials have been explored extensively in the last decade. Tissue Engineering discipline has been widely explored to provide answers associated with problems encountered in the interaction of the surgical mesh with the human body. These structures present several advantages, such as high specific surface area for cell addition, higher microporous structure and a 3D micro setting for cell–cell and cell–biomaterial interaction, associated with unique physical and mechanical properties. (8)
Synthetic non-degradable, medium or light weight are recommended for inguinal hernias when doing TAPP or TEPP. New designs like 3Dmax®, which are precut to simulate the inguinal area and ProGrip®, self-fixating mesh, are being used more often in laparoscopic inguinal repairs.
Bibliography:
- Pauli EM, Rosen MJ. Open ventral hernia repair with component separation. Surg Clin North Am. 2013;93:1111–33.
- Nguyen MT, Berger RL, Hicks SC, et al. Comparison of outcomes of synthetic mesh vs suture repair of elective primary ventral herniorrhaphy: A systematic review and meta-analysis. JAMA Surg. 2014;149:415–21.
- Peralta R, Latifi R. Long-term outcomes of abdominal wall reconstruction. what are the real numbers? World J Surg. 2012;36:534–8
- Bellín JM, Rodríguez M, García-Honduvilla N, Pascual G, Gómez Gil V, Buján J. Peritoneal effects of prosthetic meshes use to repair abdominal wall defects: monitoring adhesions by sequential laparoscopy. J Laparoend Tech. 2007; 17: 160–166.
- FitzGerald JF, Kumar AS. Biologic versus synthetic mesh reinforcement: what are the pros and cons? Clin Colon Rectal Surg. 2014; 27:140–148.
- Atema JJ, de Vries FEE, Boermeester MA. Systematic review and meta-analysis of the repair of potentially contaminated and contaminated abdominal wall defects. Am J Surg. 2016;212:982–995.
- Peppas G, Gkegkes ID, Makris MC, Falagas ME. Biological mesh in hernia repair, abdominal wall defects, and reconstruction and treatment of pelvic organ prolapse: A review of the clinical evidence. Am Surg. 2010;76:1290–9.
- Popat, K. Nanotechnology in Tissue Engineering and Regenerative Medicine, 1st ed.; CRC Press: Boca Raton, FL, USA, 2010.
Carlos Ortiz-Ortiz. MD. FACS
Celebration Center for Surgery, Florida Hospital Celebration Health, Florida. 34747.
Email: zurdo_jc@hotmail.com